Like different kids with Hunter syndrome, Cole is poor in an enzyme required to interrupt down sure molecules. Over time, toxins accumulate, and the genetic dysfunction ravages kids’s organs, together with their coronary heart — and in lots of circumstances, their mind, resulting in dementia-like signs. The situation, additionally referred to as mucopolysaccharidosis sort II, or MPS II, impacts about 500 individuals within the U.S., practically all of them boys.
Specialists consider the newly permitted drug, an intravenous enzyme substitute remedy manufactured by Denali Therapeutics, might be a recreation changer — particularly as a result of the present commonplace of care slows solely the bodily elements of the sickness. Denali’s drug additionally targets cognitive decline.
The brand new drug won’t reverse regressions which have already occurred. However it may lengthen kids’s lives and forestall many signs from displaying up for individuals who obtain it early.
“If we take a baby, very younger, and may deal with them prior to break, now the potential is sort of limitless,” stated Dr. Joseph Muenzer of the Muenzer MPS Analysis and Remedy Heart on the College of North Carolina at Chapel Hill, which sees kids with Hunter syndrome and different uncommon mucopolysaccharide illnesses.
“We don’t know the way nicely they’ll do sooner or later, however they’ll do dramatically completely different than they’d have in any other case,” he stated.
Earlier than he began to regress, Stephens’ son, Cole, was studying to learn and will speak in full sentences. Because the illness set in, talking grew to become troublesome: He may string collectively a couple of phrases, after which say only one phrase — “Mommy” — earlier than he grew to become totally nonverbal. Regardless of now being a youngster, he’s extra like a 3-year-old developmentally, Stephens stated.
The FDA’s approval of the Denali drug was a welcome shock not simply to households of kids with Hunter syndrome, however to the uncommon illness group as a complete. In current months, the FDA has come beneath hearth for rejecting a string of promising therapies for uncommon illnesses, prompting affected person advocates to stage a mock funeral with a coffin outdoors of company headquarters and triggering an investigation by Sen. Ron Johnson, R-Wis., who, previous to the Denali approval, accused the FDA of “on the lookout for excuses to say no” to therapies.
In an e-mail to NBC Information, the FDA stated the variety of approvals and rejections beneath this administration “are in keeping with historic knowledge over the past decade.” It pointed to an announcement from FDA Commissioner Dr. Marty Makary, who referred to as the Denali approval a “milestone day for kids and their households battling Hunter syndrome.” He added, “We’ll proceed to do all the pieces we are able to to speed up therapies for uncommon illnesses.”
Those that have watched kids endure from Hunter syndrome and different uncommon illnesses are hopeful that would be the case, together with Muenzer, who was a principal investigator for the Denali trial.
“These are horrible issues,” Muenzer stated. “Simply because they’re a minority doesn’t imply we should always ignore them.”
Denali’s drug, referred to as Avlayah, is the primary FDA-approved therapy within the U.S. in 20 years for Hunter syndrome — and the primary one which penetrates the blood-brain barrier, enabling it to halt the neurologic problems of the illness.
Specialists like Muenzer consider Avlayah has the potential to increase life expectancy based mostly on promising knowledge from a scientific trial that confirmed that after 24 weeks, the degrees of a key biomarker in cerebrospinal fluid related to the illness had been diminished a lot, 93% of pediatric individuals had ranges akin to people with out Hunter syndrome.
For a lot of households of kids with the situation, the approval of the drug meant extra than simply an thrilling new therapy. It additionally meant recognition of their kids’s worth.
In New Berlin, Wisconsin, 6-year-old Roran Jaskulski was identified with Hunter syndrome when he was 4. He has at all times been nonverbal, and his mom, Kylie Jaskulski, stated she worries that as a result of Roran can’t communicate, individuals who don’t know him could query if he has something to supply to others — a heartbreaking mischaracterization of her energetic, affectionate little one who likes to cuddle whereas he falls asleep and excitedly runs into faculty every day.
“He brings a lot pleasure,” Jaskulski stated. “He brings happiness and peace to each individual he interacts with.”
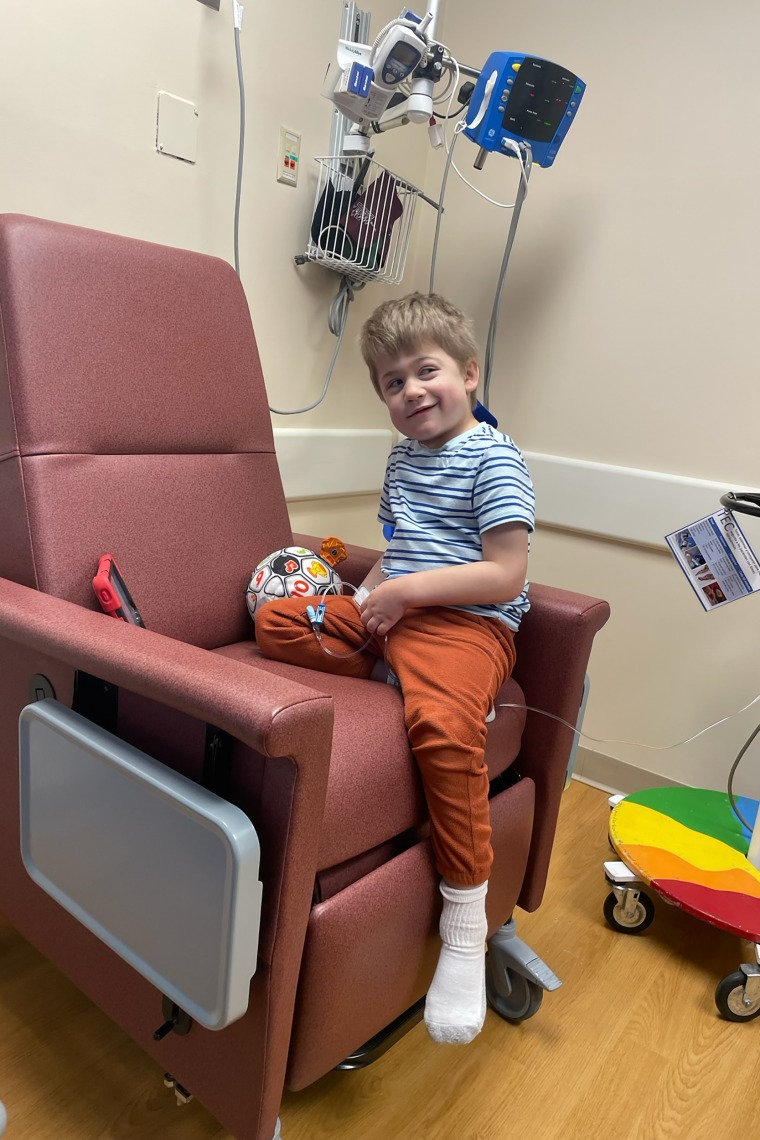
Jaskulski stated at first, receiving Roran’s prognosis felt just like the worst day of her life. However as time went on, she realized the powerlessness to stop her son from deteriorating was much more torturous.
If her insurance coverage approves the Denali drug for Roran, she stated, “possibly I don’t simply have to face by and watch.”
Like different kids with Hunter syndrome, Roran receives a weekly infusion of the present commonplace of care, a drug referred to as Elaprase permitted by the FDA in 2006. The infusions assist to stabilize his bodily decline, although not totally: In current months, Jaskulski has seen Roran growing weak spot and ache on the left aspect of his physique, affecting his gait. He additionally has gentle listening to loss.
A patchwork of states from California to Rhode Island embody Hunter syndrome on new child screenings, with extra states anticipated to begin testing for it at delivery sooner or later. Which means kids may have a greater shot at stopping cognitive impairment in the event that they take the brand new drug early, docs say.
Most Hunter syndrome youngsters have the extreme type of the illness, which has a life expectancy of between 10 and 20 years. These with the non-neurological type of Hunter syndrome, which doesn’t considerably have an effect on the mind, can stay into maturity, although they nonetheless face progressive bodily issues, primarily of their airways and coronary heart.

The prospect of the brand new therapy is thrilling for households of children on each ends of the illness. In Newkirk, Oklahoma, Christina Coldwell’s 3-year-old grandson Kashton Estes has Hunter syndrome with out cognitive issues, and he receives Elaprase infusions, which he refers to as “going to get his Spider-Man juice.” The therapy has been profitable: Coldwell stated the little boy “talks up a storm,” runs, jumps and rides a bicycle.
Nonetheless, she is raring to get Kashton on the brand new Denali drug. Two of his cousins even have the genetic dysfunction, Coldwell stated, and the household desires to stave off future well being issues in all three youngsters.
“We’re not asking for a lot: Simply give us the medication to maintain our kids alive,” she stated.
When requested about insurance coverage protection for its drug — which has an inventory worth of $5,200 per 150-milligram vial — Denali Therapeutics stated it has had “constructive” discussions with payers and stated enabling Hunter syndrome households to have fast entry to Avlayah is a “high precedence.” It additionally stated it’s seeking to increase the drug’s scientific proof for younger adults, since in the mean time, it’s only indicated for pediatric sufferers. Denali additionally stated that it goals to make use of its blood-brain barrier know-how for a variety of different situations, together with different neurodegenerative illnesses.
Stephens, the mom of 15-year-old Cole, can’t wait to begin her son on the drug. She has devoted her life to serving to not solely Cole, however others like him: In 2022, she grew to become the chief director at Muenzer’s MPS analysis and therapy middle at UNC-Chapel Hill.
When information broke final week that the FDA had granted accelerated approval to the brand new drug, Stephens ran by way of the therapy middle to inform sufferers and workers. Everybody hugged and cried.
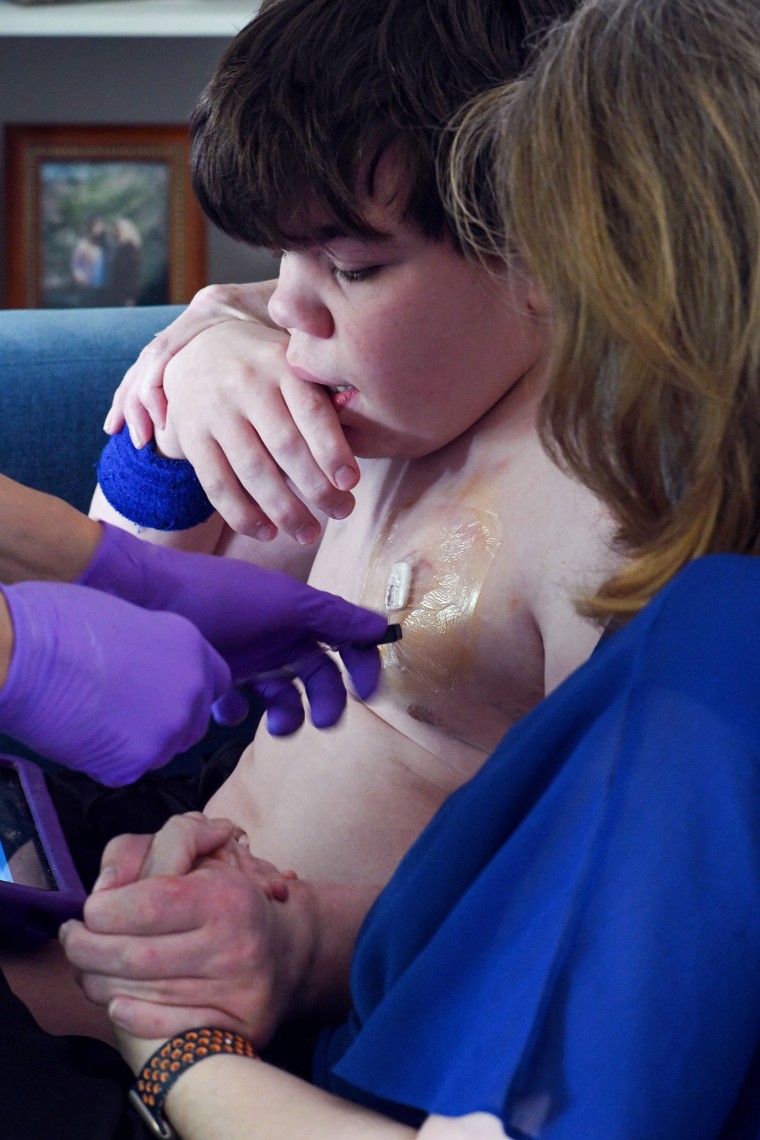
Stephens is aware of Denali’s drug can’t undo the regressions Cole already has. It received’t allow him to go to varsity or to enter the workforce. However she stated she’s nonetheless extremely grateful for it.
“My hope is he stays secure,” Stephens stated. “Secure with a progressive illness is a win.”

