Cryo-electron microscopy (Cryo-EM) has essentially reworked our understanding of the molecular equipment that governs life. For many years, structural biology was typically restricted by the “crystallization bottleneck” of X-ray diffraction. Cryo-EM modified the paradigm, permitting scientists to see into the microscopic world with unprecedented readability and element.
With its origins rooted within the pioneering work of Nobel laureates Jacques Dubochet, Joachim Frank, and Richard Henderson, Cryo-EM has developed from a distinct segment specialty—as soon as jokingly known as “blobology”—right into a mainstream powerhouse. In the present day, it’s able to revealing the hidden buildings of proteins, viruses, and mobile parts at atomic decision.
What’s Cryo-EM?
Cryo-EM is an umbrella time period for a set of superior imaging strategies used to elucidate the three-dimensional (3D) construction of organic molecules and complexes of their native, hydrated state.
On this method, the pattern is quickly frozen to temperatures under -150 °C, trapping it in vitreous ice. By imaging these frozen samples from totally different angles utilizing a transmission electron microscope (cryo-TEM), researchers seize “snapshots” of molecules as they exist in resolution. Superior computational algorithms then digitally recombine these 2D snapshots right into a high-fidelity 3D reconstruction.
The Core Precept: Vitrification
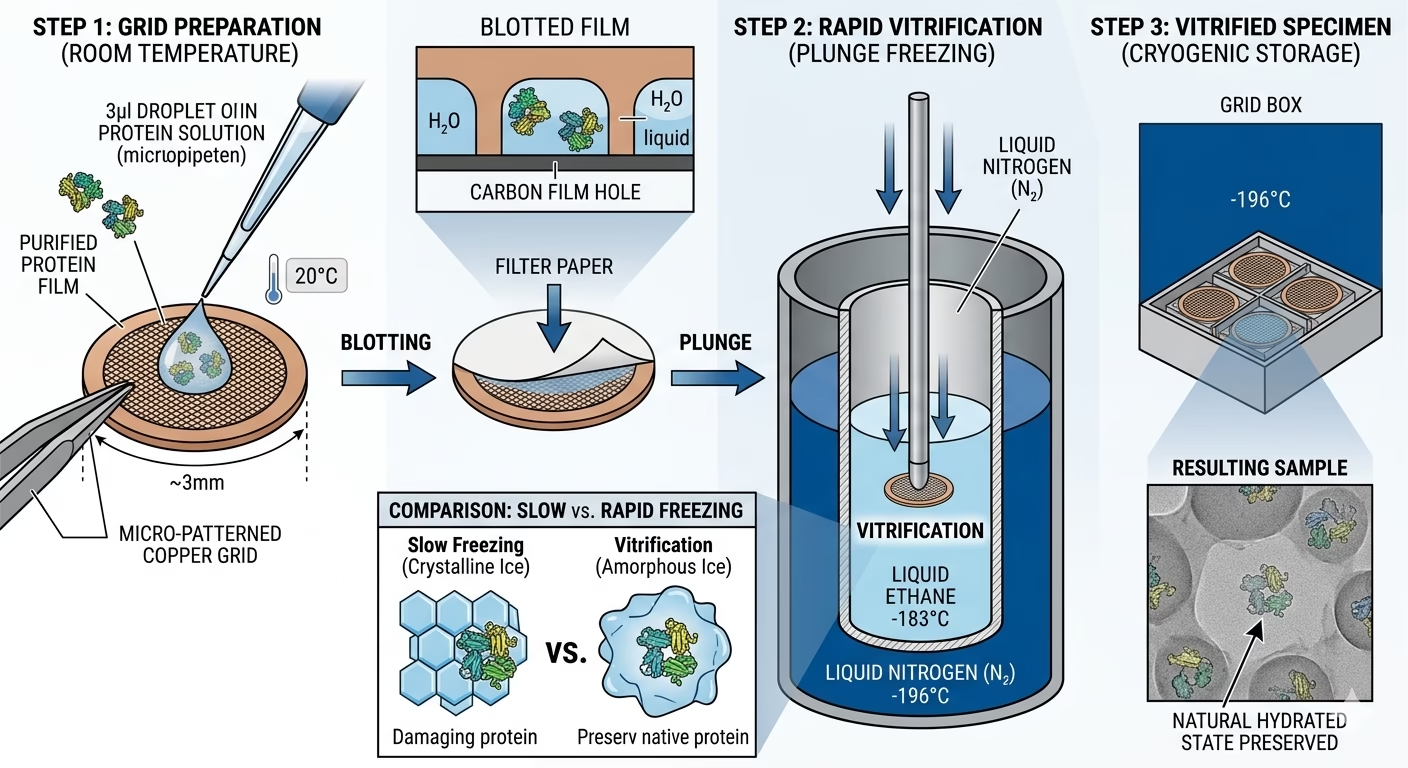
The organic relevance of Cryo-EM hinges on Vitrification. In standard electron microscopy, cooling water normally results in the formation of crystalline ice, which expands and destroys delicate protein buildings. Moreover, crystalline ice causes robust electron diffraction that dramatically reduces decision.
Vitrification entails flash-freezing the aqueous resolution so shortly (inside milliseconds) that the water molecules wouldn’t have time to crystallize. As an alternative, they type a vitreous stable—a glass-like state that’s clear to electrons. This course of offers a novel benefit: it preserves the specimen in its near-native state, offering physiologically related observations that strategies requiring harsh chemical remedies, staining, or dehydration can not match.
Historic Improvement and the Nobel Path
The event of Cryo-EM was a direct response to the constraints of standard EM, particularly pattern destruction attributable to high-energy electron beams and artifacts derived from chemical fixation.
Nineteen Seventies: Richard Henderson and Nigel Unwin produced the primary 3D construction of a organic specimen (bacteriorhodopsin) utilizing EM.
Nineteen Eighties: Jacques Dubochet developed the methodology for vitrifying water, permitting organic samples to be ready for vacuum situations with out shedding their construction.
Nineteen Nineties: Joachim Frank developed the computational methods to course of 2D photographs of randomly oriented particles into 3D buildings.
2013–Current: The “Decision Revolution” started with the event of Direct Electron Detectors and improved software program, resulting in the 2017 Nobel Prize in Chemistry.
Comparability: Standard EM vs. Cryo-EM
| Function | Standard EM | Cryo-EM |
| Pattern Prep | Fixation, staining, dehydration | Vitrification (Flash-freezing) |
| Integrity | Excessive danger of artifacts | Native construction preserved |
| Decision | Nanometer (nm) | Close to-atomic (Å) |
| Pattern Quantity | Bigger quantities required | Minimal (µg/mL) |
The Three Pillars of Cryo-EM Strategies
Cryo-EM will not be a single technique however a set of analytical instruments tailor-made to the character of the pattern.
1. Single Particle Evaluation (SPA)
The preferred Cryo-EM method and the first different to X-ray crystallography.
The Course of: Purified proteins are vitrified, leading to hundreds of molecules oriented randomly inside the ice.
The Objective: The TEM collects 2D snapshots of each doable orientation. These are then digitally aligned, categorised, and averaged right into a 3D reconstruction.
Key Achievement: SPA has just lately resolved buildings just like the human GABA-A receptor at 1.7 Å, revealing binding pockets for small molecules like histamine.
2. Cryo-Electron Tomography (Cryo-ET)
Whereas SPA appears at “purified components,” Cryo-ET appears on the “entire machine” contained in the cell.
The Course of: Used for entire cells or tissues. Since cells are thick, researchers use Targeted Ion Beam (FIB) milling to shave away layers, creating a skinny “window” or lamella.
The Objective: The pattern is tilted at numerous angles ($+/- 60) to take a sequence of 2D photographs (a tomographic tilt sequence) which can be reconstructed right into a 3D dataset referred to as a tomogram.
3. Microcrystal Electron Diffraction (MicroED)
MicroED is the “bridge” between crystallography and EM.
The Course of: It makes use of electron diffraction quite than imaging.
The Benefit: Electrons work together with matter 10^4 to 10^6 occasions extra strongly than X-rays. This permits atomic particulars to be extracted from nanocrystals (<200 nm) which can be too small for conventional X-ray diffraction.
How Cryo-EM Works: The Technical Workflow
The trendy Cryo-EM workflow has developed from a handbook, labor-intensive course of into an more and more automated pipeline. The transition from a purified pattern to a high-resolution 3D density map entails 4 vital levels:
1. Pattern Preparation and Grid Loading
The method begins with a purified macromolecule suspended in an optimized aqueous buffer. A tiny quantity (usually 3–5 microliters) is utilized to a specialised EM grid—normally a 3mm copper mesh coated with a skinny, holey carbon or gold movie.
Vital Issue: The focus have to be exactly tuned to make sure that particles are densely packed inside the grid holes however don’t overlap, which might complicate the later levels of particle selecting.
2. Vitrification: The Artwork of Flash-Freezing
The grid is mechanically “blotted” with filter paper to take away extra liquid, leaving a skinny movie of water only some hundred nanometers thick. The grid is then quickly plunged right into a main coolant, usually liquid ethane (maintained at approx. -183°C).
3. Excessive-Decision Information Acquisition (Imaging)
The vitrified grids are transferred into the high-vacuum surroundings of a Cryo-TEM (Transmission Electron Microscope) or Cryo-SEM (Scanning Electron Microscope).
Cryo-TEM: The electron beam passes by means of the pattern to disclose inside atomic buildings. That is the first software for Single Particle Evaluation.
Direct Electron Detectors (DEDs): Trendy microscopes use CMOS-based DEDs that act like high-speed cameras, capturing “films” quite than static photographs. This permits the software program to trace and proper for the motion of the pattern through the publicity.
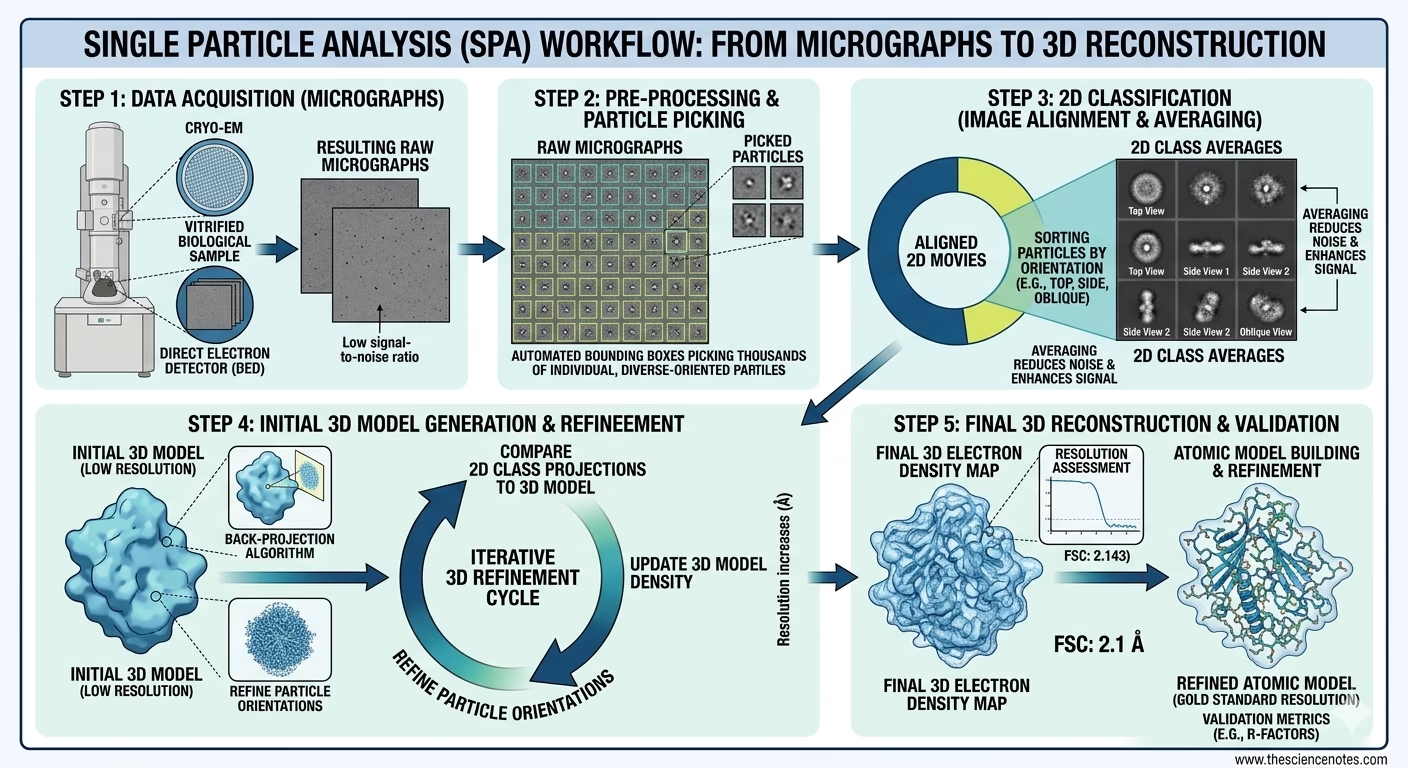
4. Computational Picture Processing
As soon as terabytes of uncooked “films” are collected, they bear rigorous digital evaluation:
Movement Correction: The software program aligns particular person film frames to repair “beam-induced movement”—the slight motion brought about when electrons strike the frozen pattern.
CTF Estimation: Calculating the Distinction Switch Perform to right for lens aberrations and defocus, making certain the ultimate map is sharp and correct.
Particle Choosing: Automated algorithms (typically powered by Deep Studying/AI) determine and extract a whole bunch of hundreds of particular person protein projections from the micrographs.
2D Classification: These projections are sorted into teams based mostly on their orientation. “Unhealthy” particles (broken proteins or contaminants) are discarded right here.
3D Refinement: By iterative back-projection algorithms, the 2D lessons are merged right into a 3D density map. This map is refined till the decision permits for the constructing of an atomic mannequin.
Arithmetic in Information Evaluation
To realize atomic decision, Cryo-EM depends on complicated sign processing to beat low distinction.
The Distinction Switch Perform (CTF)
The microscope optics introduce section shifts described by the CTF:
CTF(ok) = A(ok) sin [ π Δz λ k² + ½ π Cs λ³ k⁴ ]
The place:
Fourier Shell Correlation (FSC)
The standard of a 3D map is set by the FSC. The info is break up into two unbiased halves; the decision is outlined on the level (usually the 0.143 threshold) the place the correlation between the 2 maps drops considerably.
Strengths, Limitations, and Comparisons
Cryo-EM serves as a strong, complementary method to different structural biology strategies comparable to Nuclear Magnetic Resonance (NMR) and X-ray Crystallography.
Strengths
No Crystallization Required: Eliminates the only greatest bottleneck in structural biology, notably for giant complexes and membrane proteins.
Visualizes Giant, Versatile Complexes: Excels at resolving large molecular machines (like ribosomes) which can be too complicated for NMR.
Captures Conformational Heterogeneity: Not like the static “common” of a crystal, Cryo-EM can seize a protein “in movement,” exhibiting a number of useful states in a single pattern.
Limitations
The Small Protein Barrier: Molecules smaller than < 50 kDa are at the moment tough to picture and align attributable to their low signal-to-noise ratio.
Excessive Monetary Barrier: A top-tier setup represents a major funding, with tools prices starting from $5 to $7 million USD, plus excessive annual upkeep.
Huge Information Demand: A single session can generate a number of terabytes of uncooked information, necessitating strong GPU-based computational clusters for processing and storage.
Purposes of Cryo-EM
Structural Biology: Understanding protein capabilities and mechanisms of membrane proteins (GPCRs).
Virology: Aiding vaccine growth by visualizing viral capsids and spike proteins (e.g., SARS-CoV-2).
Drug Discovery: Rational drug design for ion channels and enormous macromolecular assemblies.
Neurobiology: Learning synaptic vesicles and protein aggregates in neurodegenerative illnesses like Alzheimer’s.
Biochemistry: Mapping the equipment of DNA replication, transcription, and translation.
Conclusion
Cryo-electron microscopy has moved from a specialty software to a transformative expertise. By enabling the visualization of organic samples of their near-native state and at excessive decision, it offers unprecedented insights into the buildings and capabilities of life’s numerous biomolecular methods. Whether or not by means of Single Particle Evaluation, Cryo-Tomography, or MicroED, it permits us to investigate proteins in all their complicated, heterogeneous glory.
X-ray Crystallography: Definition, Precept, Steps, Information Evaluation, Purposes, and Limitations